June 17-19, 2002
Moss Landing, California
Kenneth S. Johnson, J. Keith Moore and Walker O. Smith
|
June 17-19, 2002 Moss Landing, California Kenneth S. Johnson, J. Keith Moore and Walker O. Smith |
Introduction
Iron Distributions
External Iron Inputs
Iron Regulation of Ecosystem Processes
Carbon and Iron Export
Conclusions
References
Appendix A - Meeting Participants
Appendix B - Agenda
Appendix C - Downloads
A US JGOFS Workshop on Iron Dynamics in the Carbon Cycle was held at the Monterey Bay Aquarium Research Institute in Moss Landing, California on June 17 to 19, 2002. The Workshop was attended by a mix of observationalists from the US JGOFS field programs, as well as other projects, and a group of modelers (Appendix A). The goals for the meeting were:
In the following report, we highlight areas where: 1) significant progress has been made and there is a reasonable understanding of the iron cycle, 2) areas of high model sensitivity, where we currently have a fair knowledge, but where improved understanding and parameterizations are needed, and 3) areas of uncertainty in models where we have a less than fair knowledge, and where substantial new effort is needed.
Perhaps the most pressing need from a large scale modeling perspective is for the development of an expanded global database of dissolved iron distributions in the oceans (Johnson et al., 1997) similar to those available for inorganic carbon and macronutrients. Model initialization and evaluation are severely impacted by our limited knowledge of dissolved iron distributions in the world ocean. There is need for new, quality controlled measurements of dissolved iron in both surface and deep ocean waters in nearly every ocean basin. These measurements should be an integral part of any new transects or repeat hydrographic surveys of the oceans.
Such a database has not been accumulated because of the difficulties in measuring the exceedingly low levels of iron present in seawater without contamination. Recently, significant progress has been made in measuring iron concentration in seawater. The remaining uncertainties were on the order of several tenths of a nmol/L during the US JGOFS program. This represents about an order of magnitude improvement in reducing uncertainty in measurements during the past decade and greatly improves our understanding of iron distributions. The improvement in measurement capability allow us to be confident, for example, that surface iron concentrations near the US JGOFS Hawaii Ocean Time series station are significantly higher than values found at the US JGOFS EqPac site (Fig. 1). Syntheses of surface ocean Al measurements made around the world, and including many US JGOFS field programs, confirm that the higher values near Hawaii result from enhanced aerosol inputs (Measures and Vink, 2000). However, the remaining uncertainties in absolute concentrations of iron that are detected by various methods are problematic when interpreting the impact of iron on ecosystem behavior (see below). A major recommendation of the Workshop is to encourage the community to work to further reduce these uncertainties in iron measurements.
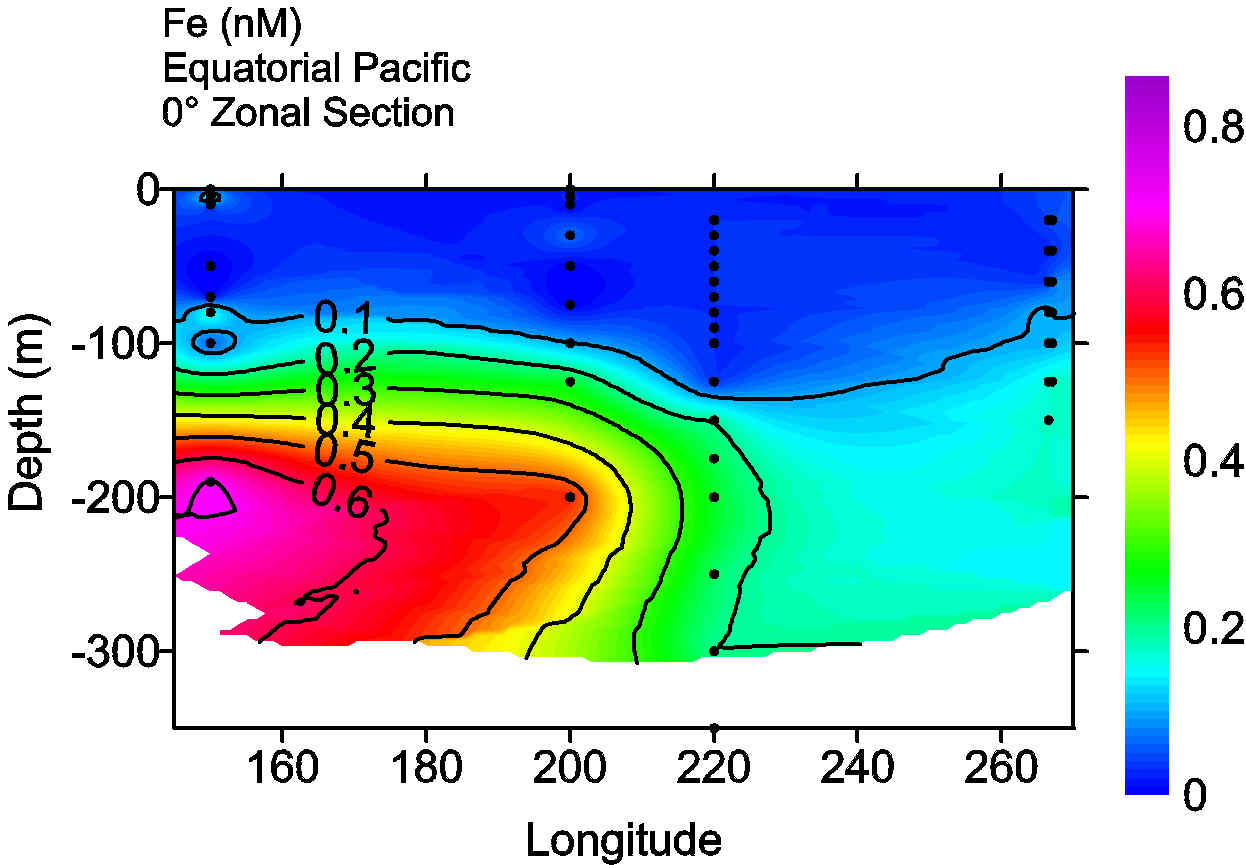
We can begin to create a global map of iron distributions by piecing together measurements from different programs, but there are considerable uncertainties in doing so. For example, one of the major results of the Equatorial Pacific iron measurements was the discovery of an iron maximum in the core of the Equatorial Undercurrent (Gordon et al., 1997; Landry et al., 1997). A sparsely populated iron section in the Equatorial Pacific can be compiled from iron measurements made in the Western Pacific during the Japanese JGOFS program (Nakayama et al., 1995), US JGOFS EqPac observations (Gordon et al., 1997) and measurements made in the Eastern Pacific during the PlumEx program (Gordon et al., 1998; this latter program was initiated with US JGOFS planning funds). This section clearly shows a plume of iron in the Undercurrent extending from the Western Pacific to the vicinity of the EqPac study site.
The elevated iron concentrations in the Equatorial Undercurrent near 150°E have been independently confirmed and attributed to a continental margin iron source (Mackey et al., 2001). Such processes are not contained in any of the global iron models. Yet these processes may have a large impact on productivity in the Equatorial Pacific (e.g. Chavez et al., 1999). The equatorial iron section (Fig. 2) also illustrates the paucity of data that is available - 4 sites that span nearly one third of the globe. A major recommendation of the workshop is that efforts begin to assemble global maps of biogeochemically significant trace element distributions, such as iron. It was concluded that, with modest additional efforts at intercalibration, such an undertaking would be feasible due to improvements in analytical and sampling technologies (e.g., Wu et al., 2001).
It is now well known that the deposition of mineral dust to the oceans is a key source of iron that modulates biogeochemical processes. Attempts to model this transport and deposition of mineral dust are increasing in complexity and accuracy (Tegen and Fung, 1994; 1995; Mahowald et al., 1999; Ginoux et al., 2001; Gao et al., 2001). There are very few measurements of dust deposition for ground-truthing these models. While the patterns of oceanic deposition predicted by these models agree qualitatively at the global scale, deposition rates in remote regions can disagree by a factor of 10 or more. More direct measurements of aerosol concentration and deposition are required, as well as the development of proxies such as aluminum concentration in surface waters. There are significant efforts to develop autonomous measurements of both of these quantities that could be deployed on moorings. Such efforts should be encouraged.
Iron flux alone, however, is not the only parameter required to assess aerosol impacts on biogeochemical processes. Solubility of iron in aerosol remains a large uncertainty with results from laboratory studies that span a range of at least an order of magnitude. Recent estimates of the amount of soluble iron within mineral dust are of 1-3 %, and well below the earlier estimates of up to 50% (Fung et al., 2000; Jickells and Spokes, 2001). Some results from field observations, which were presented at the meeting, indicate that even larger values may be observed in natural systems. Variability in aerosol solubility that has been observed in laboratory studies may also be a function of the aerosol source region, but there has been no systematic assessment. In addition, there can be large differences in the solubility of iron delivered by wet and dry deposition. These processes should be considered separately.
In addition to the large input of iron from aerosol, continental margins appear to be a large source of iron. Much of the iron is derived from sedimentary sources. Just as water upwelled in the Southern Ocean and along the Equator is iron limited, water upwelled along the coast would also be iron limited without this sedimentary source. Because the iron source need not be directly coupled to macronutrients in the source water, iron limitation may occur in the coastal zone, just as it does in open ocean waters. Presentations at the work shop demonstrated that coastal iron limitation can play a major role in regulating coastal ocean ecosystem rates, biomass and structure (Hutchins and Bruland, 1998; Hutchins et al., 1998; Johnson et al., 2001). The effects of iron near the ocean margins may have an impact on the ocean carbon cycle that is as large as the apparent effect in open ocean waters. The coastal ocean accounts for 20% of ocean primary productivity, maybe 50% of the carbon export and 90% of C burial. Iron limitation of coastal primary production may be particularly important at low stands of sea-level during the glacial maxima. Further, as shown in Fig. 2, the continental margins may be a significant iron source to some areas of the open ocean.
This continental margin source is not presently incorporated in any of the global models. However, much still remains to be determined before the influence of ocean margins can be quantitatively modeled. We do not yet have the knowledge base that would allow us to determine the quantitative offshore fluxes of iron from ocean margins. Mechanisms of sediment resuspension and transport of the sediment-bound iron from the benthic boundary layer to the euphotic zone are not incorportated in global models. The composition and bioavailability of iron within this material is not well understood. We do not understand the changes in ecosystem processes that are, perhaps, reflected in higher Fe:C ratios in coastal phytoplankton (Sunda and Huntsman, 1995). We expect that coastal ecosystems are more efficient at exporting C, as a result of higher Fe concentrations, but we cannot quantify this process. Finally, most of our understanding of iron limitation in coastal areas comes from studies of eastern boundary areas such as central California. A more global picture is needed, including western boundaries and high latitudes.
Iron Regulation of Ecosystem Processes
Very significant improvements in our understanding of the role of iron in regulating ecosystem processes have been made. Just one decade ago there were intense debates about the role of iron in controlling biogeochemical processes (e.g., Banse, 1990; Cullen, 1991; Martin et al., 1991). Since that time six open ocean iron fertilization experiments have been conducted in the Equatorial Pacific, Southern Ocean and sub-Arctic Pacific (Table 1). All have shown enhanced rates of primary production and biomass accumulation following iron addition. Iron enrichment experiments conducted in bottles during the US JGOFS programs have shown a remarkable commonality in the level of iron that stimulates a community response. Community growth rates and nutrient uptake rates in these bottle experiments can be fitted with a Michaelis-Menton (Monod) model using iron as a limiting nutrient. The half-saturation constant is approximately 0.1 nM Fe in most open ocean environments that have been examined (Fitzwater et al., 1996; Coale et al., in prep.). Values reported for experiments in the coastal zone, where iron concentrations are greater, are higher ~0.5 nM (Hutchins). Such parameterizations have formed the basis for incorporation of iron into global ecosystem models.
Future efforts must focus on experiments that provide information at the individual phytoplankton species or functional group level. This is necessary because it is apparent that the differential response of each functional group plays a role in sustaining the ecosystem, exporting carbon and altering nutrient uptake and export ratio's. Modeling ecosystems at the species and functional group level may prove problematic, though. Detailed information is required for each group and the resulting model complexity is not easy to interpret and it is difficult to apply in high resolution global simulations. There was considerable discussion regarding the need to develop simpler relationships that encompassed the diversity of functional groups and their impacts on biogeochemical processes through parameters such as plankton size.
In most modeling studies to date, iron has been treated in a manner similar to the macronutrients with the implicit assumption that iron is recycled in a similar manner and at similar rates to the macronutrients. However, iron may behave differently within phytoplankton cells (i.e. Armstrong, 1999) and within ecosystems than the major macronutrients. There is considerable uncertainty as to whether iron is recycled at similar rates within surface waters and with similar remineralization length scales at depth as organic carbon and the macronutrients. Some progress has been made over the last decade in estimating the Fe/C ratios of different biotic pools in marine systems (summarized in Price and Morel, 1998). There is very little data about how these ratios may vary over space and time, between laboratory and in situ settings, and of the key differences between coastal and open ocean systems. The available data suggests that Fe/C ratios are more variable than N/C or N/P ratios in marine systems.
Detailed measurements of key elemental ratios (Fe/C/N/P/Si/CaCO3) using clean techniques within the various biotic, dissolved, and detrital pools in surface waters and within sinking particulate matter throughout the water column could answer many of our remaining questions about iron and carbon cycling in the oceans, and should be a high priority in the future. Such experiments must provide information at the individual phytoplankton species or functional group level. This is necessary because it is apparent that the differential response of each functional group plays a role in sustaining the ecosystem, exporting carbon and altering nutrient uptake and export ratio's. Preliminary results described at the meeting from the AESOPS program, as well as data collected in other JGOFS experiments, demonstrate that it is feasible to collect such information. A sufficiently large data set has not been obtained that would allow broad generalizations.
Many of the discussions at the workshop turned on iron speciation and the role of ligands, and how these processes may influence bioavailability and biogeochemical cycling in the oceans. A consensus of the meeting was "probably all iron is bioavailable, but we don't understand the time scales or processes". Phytoplankton and bacteria have a complex array of Fe acquisition systems at their disposal - siderophore mediated uptake (Granger and Price, 1999), reductases (Maldonado and Price, 2001), ligand production, and phagotrophy (Maranger et al., 1998). Processes such as photochemistry clearly cause redox cycling of iron between different chemical species, thereby increasing bioavailability. There also appears to be some degree of species specificity in the forms of iron accessed by prokaryotes vs. eukaryotes (Hutchins et al., 1999). Such processes are not generally included in models. Further, it is recognized that organisms can increase their iron uptake capacity in response to iron stress, and can also engage in luxury iron uptake. These findings need further study and clarification. They potentially have large implications for the role of iron speciation influence on phytoplankton community composition and biogeochemical cycling in surface waters. It is possible that we need to develop mechanistic models of Fe speciation, photochemistry and uptake at the cell surface as a sensitivity analysis tool to delineate the relevant uncertainties which could guide further experiments and larger scale modeling efforts.
It is now known that >99% of the dissolved iron in the upper ocean is strongly complexed by organic molecules (Rue and Bruland, 1995; Wu and Luther, 1995). These ligands are distributed more or less uniformly in the oceans (within a factor of 2-3, based on very few measurements). Currently, there is not enough known about the ligands which bind iron and help keep it in solution in oceanic waters to include them within models, except in the very crude ways. Many marine micro-organisms are capable of producing iron binding ligands. It is clear that there is more than one class of iron binding ligand, but we are less certain whether these ligands comprise two broad classes of complexing molecules or a continuum of complexing strengths. The sources and sinks, lifetimes, and turnover rates between ligand pools and the processes which govern these transformations (biology vs. photochemistry) of ligand pools are simply unknown. We need to know the direct sources of these ligands and their predominant removal processes (particularly if ligand removal controls iron removal). We need to understand the size classes of these ligands and their distributions in space and time. It is not clear yet if Fe(II) stabilizing ligands play an important role in iron chemistry. Finally, we need to understand the impacts of photochemistry in regulating turnover of ligands.
Great progress in understanding the links between nitrogen fixation and marine iron cycling have been made in recent years through field and laboratory observations, satellite data analysis, and modeling efforts. It has been demonstrated that the iron requirements for Trichodesmium spp., a key nitrogen fixing cyanobacterium, are approximately 10-fold higher than for most open ocean phytoplankton with an iron-replete Fe/C ratio of ~50 µmol/mol (Frank-Berman et al., 2001; Kustka et al., submitted). Nitrogen fixing diazotrophs, largely modeled using observations on Trichodesmium have begun to be incorporated into marine ecosystem models (Hood et al., 2001; Fennel et al., 2002; Moore et al., 2002a). Recent fieldwork has presented strong evidence of phosphorus limitation of nitrogen fixation in parts of the North Atlantic (where dust inputs are high) (Wu et al., 2001; Sañudo-Wilhelmy et al., 2001). Satellite data analysis and modeling studies suggest that iron and/or light may be limiting nitrogen fixation rates over much of the rest of the tropical/subtropical ocean (Frank-Berman et al., 2001; Moore et al., 2002b).
We have a good, basic understanding of the role of iron in stimulating the onset of phytoplankton blooms that has been developed through bottle experiments and open ocean iron fertilizations. However, we have a much poorer understanding of the fate of carbon produced in the open ocean iron enrichment experiments. Carbon is not exported in bottle experiments and open ocean iron fertilizations have not yet clearly seen the bloom termination. It is not apparent, therefore, what regulates carbon export. Discussions at the meeting suggested that small scale (~10 km) fertilization experiments may not achieve the high biomass conditions conducive to carbon export. Horizontal diffusion appears to dilute the patches with unfertilized, low biomass waters and particle concentrations do not reach a point where aggregation and sinking occurs. Such understanding is critical to the development of linked iron and carbon models. A major recommendation of the Workshop was that future open ocean iron fertilization experiments be conducted in environments such as the Equatorial Pacific where carbon export is easier to observe and that the experiments be of sufficient size to enhance the likelihood of observing the conditions that lead to export.
The workshop discussions also highlighted how little we understand the processes by which dissolved iron is removed from the upper ocean by adsorption and scavenging by particles. This sink for dissolved iron is critical for removing dissolved iron from the upper ocean and maintaining sub-surface dissolved iron concentrations at depleted values relative to the macronutrients in terms of the nutritional demands of the phytoplankton. Large amounts of dissolved iron enter surface waters each year from mineral dust depostion. Biological uptake and removal through sinking particulates can remove only a fraction of this new iron. The rest must be removed through adsorption/scavenging processes. Little is known about how this key loss process for dissolved iron varies with particle concentration and composition, sinking particulate flux, iron concentrations, or about its reversability. Current modeling efforts often assume that scavenged iron is permanently lost to the sediments. However, the elevated iron concentrations in the deep ocean beneath the high dust deposition regions suggests that there is considerable remineralization of iron adsorbed onto sinking particles within the water column. The relatively constant iron concentrations in the deep ocean (varying at most by a factor of 3-4) suggest that adsorption/scavenging losses must be much lower than in surface waters. How this surface/deep ocean pattern is driven by ligand dyanamics, iron concentrations, and particle dyanmics merits significant further study. Understanding deep ocean iron cycling is critical for modeling ocean biogeochemistry over long timescales.
There have been tremendous advances in our understanding of iron cycling in the ocean during the past ten to fifteen years, and we now recognize iron as a keystone regulator of biogeochemical functioning. However, it is also clear that the chemistry of iron can be exceedingly complex. Further, it is unlikely that complex models of iron cycling with many, poorly constrained parameters will lead to successful, prognostic models and a predictive understanding of the effects of iron on ocean biogeochemistry.
A clear message from the modeling community at the meeting was for parameterizations that might lead to relatively simple equations of iron chemistry and ecosystem response to iron concentration. At first glance, such requests might appear to be wishful thinking. However, many of the areas where we have developed a reasonable database of reliable observations do lend themselves to simple parameterizations. One example is the use of Michaelis-Menton equations to predict the impact of iron on community growth rates. Clearly much additional work remains to be done, particularly at the species and functional group level. The key question now is whether additional work will continue to support such basic parameterizations, perhaps with extensions based on additional parameters such as cell size, or whether additional work demonstrates that the system is so complicated that simple models will be of little use.
Armstrong, R.A., 1999. An optimization-based model of iron-light-ammonium colimitation of nitrate uptake and phytoplankton growth. Limnol. Oceanogr., 44, 1436-1446.
Banse, K., 1990. Does iron really limit phytoplankton production in the offshore subarctic Pacific? Limnol. Oceanogr., 35, 772-775.
Berman-Frank, I., Cullen, J.T., Shaked, Y., Sherrell, R.M., Falkowski, P.G., 2001. Iron availability, cellular iron quotas, and nitrgen fixation in Trichodesmium. Limnol. Oceanogr., 46, 1249-1260.
Chai, F., Lindley, S.T., Barber, R.T., 1996. Origin and maintenance of a high nitrate condition in the equatorial Pacific. Deep-Sea Research II, 43, 1031-1064.
Chavez F.P., Strutton P.G., Friederich C.E., Feely R.A., Feldman G.C., Foley D.C., McPhaden M.J., 1999. Biological and chemical response of the equatorial Pacific Ocean to the 1997-98 El Nino. Science 286, 2126-2131.
Christian, J.R., Verschell, M.A., Murtugudde, R., Busalacchi, A.J., McClain, C.R., 2002a. Biogeochemical modelling of the tropical Pacific Ocean I. Seasonal and interannual variability. Deep-Sea Research II, 49, 509-543.
Christian, J.R., Verschell, M.A., Murtugudde, R., Busalacchi, A.J., McClain, C.R., 2002b. Biogeochemical modelling of the tropical Pacific Ocean II. Iron biogeochemistry. Deep-Sea Res. II, 49, 545-565.
Cullen, J. J., 1991. Hypotheses to explain high-nutrient conditions in the open sea. Limnol. Oceanogr. 36, 1578-1599.
Fennel, K., Spitz, Y.H., Letelier, R.M., Abbott, M.R., 2002. A deterministic model for N2-fixation at station ALOHA in the subtropical North Pacific. Deep-Sea Res. II, 49, 149-174.
Fitzwater, S. E., K. H. Coale, R. M. Gordon, K. S. Johnson and M. E. Ondrusek. 1996. Iron deficiency and phytoplankton growth in the Equatorial Pacific. Deep-Sea Research II, 43 995-1015.
Fung, I.Y., Meyn, S.K., Tegen, I., Doney, S.C., John, J.G., Bishop, J.K.B., 2000. Iron supply and demand in the upper ocean. Global Biogeochem. Cycles, 14, 281-291.
Gao, Y., Kaufman, Y.J., Tanre, D., Kolber, D., Falkowski, P.G., 2001. Seasonal distributions of aeolian iron fluxes to the global ocean. Geophys. Res. Lett., 28, 29-32.
Ginoux, P., Chin, M., Tegen, I., Prospero, J.M., Holben, B., Dubovik, O., S. Lin, 2001. Sources and distributions of dust aerosols simulated with the GOCART model. J. Geophys. Res.,106, 20255-20273.
Gordon, R. M., K. H. Coale and K. S. Johnson. 1997. Iron distributions in the Equatorial Pacific, implications for new production. Limnology and Oceanography 42, 419-431.
Gordon, R. M., K. S. Johnson and K. H. Coale. 1998. The behavior of iron and other trace elements during the IronEx I and PlumEx experiments in the Equatorial Pacific. Deep-Sea Research II, 45, 995-1041.
Granger J., Price N.M., 1999. The importance of siderophores in iron nutrition of heterotrophic marine bacteria. Limnology and Oceanography 44, 541-555.
Hood, R.R., Bates, N.R., Capone, D.G., Olson, D.B., 2001. Modeling the effect of nitrogen fixation on carbon and nitrogen fluxes at BATS. Deep-Sea Res. II, 48, 1609-1648.
Hutchins D.A., Bruland K.W., 1998. Iron-limited diatom growth and Si , N uptake ratios in a coastal upwelling regime. Nature 393, 561-564.
Hutchins D.A., DiTullio G.R., Zhang Y., Bruland K.W., 1998. An iron limitation mosaic in the California upwelling regime. Limnology and Oceanography 43, 1037-1054.
Hutchins D.A., Witter A.E., Butler A., Luther G.W., 1999. Competition among marine phytoplankton for different chelated iron species. Nature 400, 858-861.
Hutchins, D.A., Witter, A.E., Butler, A. and Luther, G.W. III. 1999. Competition among marine phytoplankton for different chelated iron species. Nature 400, 858-861.
Jickells, T., Spokes, L., 2001. Atmospheric iron inputs to the oceans. In, Turner, D., Hunter, K. (Eds.) Biogeochemistry of Iron in Seawater. Wiley, pp. 85-121.
Johnson K., Chavez F.P., Elrod V.A., Fitzwater S.E., Pennington J.T., Buck K.R., Walz P.M., 2001. The annual cycle of iron and the biological response in central California coastal waters. Geophysical Research Letters 28, 1247-1251.
Johnson K.S., Gordon R.M., Coale K.H., 1997. What controls dissolved iron concentrations in the world ocean? Marine Chemistry 57, 137-161.
Lancelot, C., Hannon, E., Becquevort, S., Veth, C., De Baar, H.J.W., 2000. Modeling phytoplankton blooms and carbon export production in the Southern Ocean, dominant controls by light and iron in the Atlantic sector in Austral spring 1992. Deep-Sea Res. I, 47, 1621-1662.
Landry, M. R. et al., Iron and grazing constraints on primary production in the central equatorial Pacific, an EqPac synthesis. Limnol. Oceanogr., 42, 405-418, 1997.
Leonard, C.L., McClain, C.R., Murtugudde, R., Hofmann, E.E., Harding Jr., L.W., 1999. An iron-based ecosystem model of the central equatorial Pacific. J. Geophys. Res., 104, 1325-1341.
Loukos, H., Frost, B., Harrison, D.E., Murray, J.W., 1997. An ecosystem model with iron limitation of primary production in the equatorial Pacific at 140°W. Deep-Sea Res. II, 44, 2221-2249.
Mackey, D. J., O'Sullivan, J. E., Watson, R. J. 2002. Iron in the Western Pacific, a riverine or hydrothermal source for iron in the Equatorial UnderCurrent? Deep-Sea Res. I, 49, 877-893.
Mahowald, N., Kohfeld, K., Hansson, M., Balkanski, Y., Harrison, S.P., Prentice, I.C., Schulz, M., Rodhe, H., 1999. Dust sources and deposition during the last glacial maximum and current climate, A comparison of model results with paleodata from ice cores and marine sediments. J. Geophys. Res., 104, 15895-15916.
Maldonado M.T., Price N.M., 2001. Reduction and transport of organically bound iron by Thalassiosira oceanica (Bacillariophyceae). J. Phycol. 37, 298-310.
Maranger R., Bird D.F., Price N.M., 1998. Iron acquisition by photosynthetic marine phytoplankton from ingested bacteria. Nature 396, 248-251.
Martin, J. H., Gordon, R. M. Fitzwater, S. E., 1991. The case for iron. Limnol. Oceanogr. 36, 1793-1802.
Measures, C. I., Vink, S. 2000. On the use of dissolved aluminum in surface waters to estimate dust deposition to the ocean, Global Biogeochem. Cycles 14, 317-327.
Moore, J.K., Doney, S.C., Glover, D.M., Fung, I.Y., 2002b. Iron cycling and nutrient limitation patterns in surface waters of the world ocean, Deep-Sea Res. II, 49, 463-508.
Moore, J.K., Doney, S.C., Kleypas, J.C., Glover,D.M., Fung, I.Y., 2002a. An intermediate complexity marine ecosystem model for the global domain, Deep-Sea Res. II, 49, 403-462.
Nakayama, E., H. Obata, K. Okamura, K. Isshiki, H. Karatani, T. Kimoto, 1995. Iron and manganese in the atmosphere and oceanic waters. In H. Sakai and Y. Nozaki, eds., Biogeochemical Processes and Ocean Flux in the Western Pacific, Terra Scientific Publishing, Tokyo, pp. 53-68.
Price N.M., Morel F.M.M., 1998. Biological Cycling of Iron in the Ocean. In, A. Sigel, H. Sigel (Eds.), Iron transport and Storage in Microorganisms, Plants, and Animals, Vol. 35, Marcel Dekker, Inc, pp. 1-35.
Rue E.L., Bruland K.W., 1995. Complexation of iron(III) by natural organic ligands in the Central North Pacific as determined by a new competitive ligand equilibration/adsorptive cathodic stripping voltammetric method. Marine Chemistry 50, 117-138.
Sañudo-Wilhelmy, S.A., Kustka, A.B., Gobler, C.J., Hutchins, D.A., Yang, M., Lwiza, K., Burns, J., Capone, D.G., Raven, J.A., Carpenter, E.J., 2001. Nature, 411, 66-69.
Sunda W.G., Huntsman S.A., 1995. Iron uptake and growth limitation in oceanic and coastal phytoplankton. Marine Chemistry 50, 189-206.
Tegen, I., Fung, I.Y., 1994. Modeling of mineral dust in the atmosphere, Sources, transport, and optical thickness, J. Geophys. Res., 99, 22897-22914.
Tegen, I., Fung, I.Y., 1995. Contribution to the atmospheric mineral aerosol load from land surface modification. J. Geophys. Res., 100, 18707-18726.
Wu J., Luther G.W., 1995. Complexation of Fe(III) by natural organic ligands in the Northwest Altantic Ocean by a competitive ligand equilibration method and a kinetic approach. Marine Chemistry 50, 159-177.
Wu, J. and G. W. Luther, Spatial and temporal distribution of iron in the surface water of the northwestern Atlantic Ocean, Geochim. Cosmochim. Acta, 60, 2729-2741, 1996.
Wu, J., Boyle, E., Sunda, W., and Wen, L.-S., 2001. Soluble and colloidal
iron in the oligotrophic North Atlantic and North Pacific. Science
293, 847-849.
Table 1. A summary of open ocean iron enrichment experiments that have been conducted to date.
| IronEx I: | Equatorial Pacific, 1993. 3-fold increase in chlorophyll. Patch subducted
4 days into the experiment.
(Martin et al., 1994, Nature 371, 123-129) |
| IronEx II: | equatorial Pacific, 1996. 10-fold increase in chlorophyll, 90 µatm
draw down in CO2, 5µM drawdown in NO3.
(Coale et al., 1996, Nature 383, 495-501) |
| SOIREE: | Pacific sector of Southern Ocean, summer 1999. South of Polar Front.
6-fold increase in chlorophyll, 25 µatm draw down in CO2,
2 µM draw down in NO3.
(Boyd et al., 2000, Nature 407, 695-702) |
| EisenEx-1: | Atlantic sector of Southern Ocean, spring 2000. Dispersion into an
eddy, 4x increase in chlorophyll.
(Strass et al., Abstract OS11L-06 AGU/ASLO 2002 Ocean Sciences Meeting, Honolulu, HI) |
| SEEDS: | western subarctic Pacific Ocean, summer 2001. 40-fold increase in chl,
13 µM draw down in NO3.
(Saito et al., Abstract OS41B-07, AGU/ASLO 2002 Ocean Sciences Meeting, Honolulu, HI) |
| SOFeX: | Pacific sector of Southern Ocean, summer 2002. N. and S. of Polar Front.
>10x increase in chlorophyll, >40 µatm draw down in CO2.
(http://www.mbari.org/education/cruises/SOFeX2002/Logbook.htm) |
Figure 1. Vertical profiles of dissolved
iron measured at the US JGOFS EqPac study site (A) reported by Gordon et
al. (1997) and at the US JGOFS Hawaii Ocean Time series (HOT) Station ALOHA
(B) (Rue and Bruland, 1995; Wu et al., 2001; Johnson et al., in prep.).
Variability in surface waters at Stn ALOHA is driven by changes in deposition
rate of aerosol iron, which produces the surface water iron maximum.
Figure 2. A vertical section of iron along
the equator in the Pacific Ocean. The section was constructed from two
iron profiles reported by Nakayama et al. (1995) at 150°E and 158°W,
EqPac measurements reported by Gordon et al. (1997) at 140°W and PlumEx
profiles reported by Gordon et al. (1998) at 93°W. Note that one additional
profile reported by Nakayama at 158°W was excluded as iron concentrations
were substantially elevated and inconsistent with other equatorial Fe profiles.
Appendix A - Meeting Participants
Rob Armstrong, SUNY Stony Brook
Kathy Barbeau, Scripps
Phil Boyd, NIWA U. Otago
Ed Boyle, MIT
Ken Buesseler, WHOI
Zanna Chase, MBARI
Francisco Chavez, MBARI
Jim Christian, UM
Jay Cullen, WHOI
John Dunne, Princeton
Katja Fennel, OSU
Steve Fitzwater, MBARI
Mike Gordon, MLML
Mike Hiscock, Duke U.
Dave Hutchins, U. Delaware
Ken Johnson, MBARI
Zbigniew Kolber, Rutgers
Adam Kustka, SUNY Stony Brook
Phoebe Lam, UC Berkeley
Chuck McClain, NASA
Chris Measures, U. Hawaii
Keith Moore, UC Irvine
Jim Murray, U. Washington
Lisa Pickell, MBARI
Peter Sedwick, BBSR
Walker Smith, VIMS
Peter Strutton, MBARI
Wendy Wang, MLML
Mark Wells, U. Maine
Jingfeng Wu, MIT
Detailed agenda is listed on Iron Workshop Site
You may download this document in other formats:
MS-WORD: iron_rpt.doc
PDF: iron_rpt.pdf