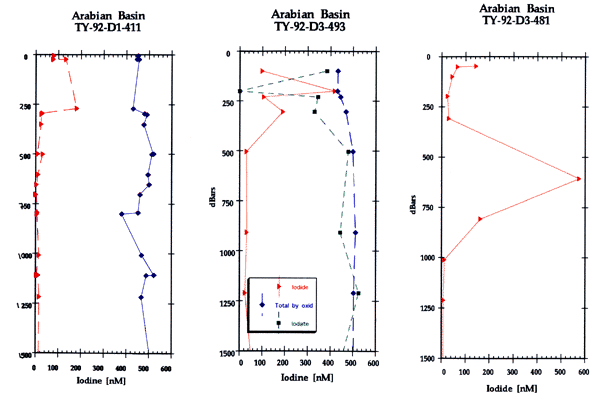
*High levels of iodide were found below the euphotic zone in the Arabian and Somali Basins during the Southwest Intermonsoon 1992 (Farrenkopf et al. in press) as part of the Netherlands Indian Ocean Programme leg D cruises.
1992 Netherland's Indian Ocean Programme (NIOP)
Since November of 1992 we have collected samples in the Arabian Basin during two additional seasons: the Northeast Intermonsoon and the Southwest Monsoon seasons of 1995. These data are the focus of this presentation. A Station map is provided for the five different cruise tracks. A more detailed map is also provided for the station intercomparison discussion.
1995 During U.S. JGOFS Process #2 (March-April) and WOCE (July)


Below, dissolved iodide concentrations at station 2 are greater than 200 nM at all depths sampled down to approximately 600 m. Nitrite is low but also detectable at the same depths (0.07 µM). At 600 m depth [I-] = 570 nM. Below 800 m [I-] < 10 nM. In distinct contrast to both Pacific and Atlantic data which traditionally show [I-] 10 nM below 200 m.

This Arabian Sea data is unique at station two in that iodate reduction is observed to be significantly greater than nitrate reduction. By station 11 where the nitrite concentrations reached their highest values (4.5 µM), [I-] is measured at values greater than 400 nM and as high as 650 nM at the nitrite maxima (250 m). The high iodide concentrations reach well into the OMZ and below 600 m. Virtually no iodate is detected. Additionally, preliminary experimental data shows the consumption of iodate added to water incubated from 200 m depth and a corresponding increase in iodide concentrations within six hours.
Experiments to isolate the size fraction in which the iodate reduction is taking place showed that samples taken from 200, 400 and 600 m (well below the euphotic zone) showed changes in speciation in the unfiltered water but not in the 0.2 µm fraction. Some change was noted in the 0.4 µm fraction.
Equipment and Methods
Iodide and iodate concentrations were determined using polarographic methods. Iodide (I-) was measured using cathodic stripping square wave voltammetry (CSSWV)[Luther, 1988 #109]. Iodate (IO3-) was measured using differential pulse polarography (DPP)[Herring, 1974 #69]. Total iodine (·Ired) was measured using CSSWV [Campos (in press)]. Methods of detection for total iodine are presently available for either the oxidation of iodide to iodate or the reduction of iodate to iodide (Takayanagi and Wong, 1986; Wong and Zhang, 1992 and Campos (in press)). In this study, the ascorbic acid reduction technique was used. The instrument minimum detection limits in seawater for I-, IO3-, and ·Ired using polarography are 0.2, 20, and 5 nM respectively. Electrochemical measurements were made in a borax buffer solution in a 10 mL polarographic cell. EG & G Princeton Applied Research model 384 B polarographic analyzers equipped with 303A hanging mercury drop electrode (HDME) stands were used throughout. Potentials were measured vs. a saturated calomel reference electrode (SCE) and a platinum counter electrode. Iodide gives rise to a peak at a potential of -0.306 V, and iodate has a peak potential of -1.08 V. Aboard ship 10.0 mL aliquots of sample were dispensed into glass voltammetric cells and the concentrations of iodine species were determined by the method of standard addition. A minimum of three standard additions were made for each determination.
Sulfide samples were drawn anaerobically via syringe at the CTD rosette and analyzed by polarography within ten minutes [Luther, 1991 #371; Farrenkopf et al. and Theberge et al., in press].
Oxygen and peroxide can be determined without deposition by applying a negative potential scan from 0.0 V to -1.5 V using the linear sweep or square wave voltammetry. Oxygen and peroxide have peaks near -0.09 V and -0.95 V, respectively (Meites, 1965).
Iodate reduction greater than Nitrate depletion
In the most western part of the Arabian basin (off the coast of Oman ), inspite of the fact there is 20-30 uM nitrate throughout the Oxygen Minimum Zone (OMZ), there is a considerable depletion of iodate. Specifically, in some instances there is MORE reduction of iodine than nitrogen. The appropriate comparison is not the "starting material" [relative concentrations of IO3- and NO3-], but the product of reduction measured in the water column as I- and NO2-.
Iodide concentrations measured in our lab are comparable and in some instances far exceed the nitrite levels at the same depths from the same stations. In the northwest and north central parts of the basin, the dissolved nitrite concentrations in the OMZ are at least a factor of four to five (and generally one to two orders of magnitude) lower than in waters in the southeastern sector. We contend that a more accurate assessment of the importance of the various redox cycles can be made by consideration of the relative magnitude of the concentration of the reduced nitrogen intermediate nitrite (as an approximation of denitrification) to the concentration of iodide (measured in this study).
It is clear from estimates of denitrification and measurements of nitrite in the Arabian Sea that reduction of nitrate may play a significant role in the oxidation of carbon in this region. Previous work has shown that the denitrification process is limited in geographical extent to waters north of 12-14°N latitude (e.g. Naqvi, 1987). The reigning assumption until recently has been that denitrification occurred throughout the entire Arabian Sea north of this boundary. Naqvi (1991) has clearly demonstrated that this assumption is incorrect. The highest rates of denitrification are observed in the eastern part of the basin, and/or near the Indian shelf, with lateral advection of nitrite toward the southwest. Naqvi (1991) compiled all the nitrite maxima data available at the Indian National Oceanographic Data Centre (INODC). This exercise clearly showed that in contrast to the southeastern part of the basin, the northwestern waters are characterized by comparatively low nitrite concentrations.
Nitrogen species measurements made during the NIOP (van Koutrik et al., 1994) and during Process Cruise #2 (Codispoti and Morrison PI's) are consistent with the conclusions of Naqvi (1991) .
Neglecting for the moment any possible differences in microbial reduction rates for the two elements, the data would suggest that the iodine redox cycling in the OMZ of the northwest and north central parts of the basin is at least of the same order of magnitude as that for nitrate cycling and may in some instances be considerably greater. The relative importance of iodine reduction may in fact be greater than the data suggest, as the observed nitrite may be the product of both denitrification and ammonia oxidation. Therefore the microbial reduction of iodate in the OMZ of the northwest Indian Ocean may play as significant a role as denitrification in the decomposition of organic matter.

Shown in this figure of Nitrite (NO2)- vs. depth for station 21 are five separate CTD casts at one location (the ship was repositioned prior to sampling). Despite differences in sampling density, a distinct change in (NO2)- over the course of a couple days is evident. This type of change in structure of the profile was observed at several stations and during previous cruises (per comm. Lou Codispoti).
Nutrient Data Courtesy of Katherine Krogslund on behalf of U.S.JGOFS Hydro crew. Lou Codispoti and John Morrison PI's
"ARABIAN SEA PARADOX"
This term is taken from the zooplankton literature (Marten Baars et al.) in reference to the consistent abundance of zooplankters regardless of local production.
High level nutrients, high production (> 1 g C m-2d-1) during the SW and intermonsoon lower nutrients, lower production (0.3 g m-2d-1) during the NE and intermonsoon. From a chemical perspective, this implies a rapid oxidation of the reduced species (time scales months). Lower oxygen and reduced species resulting from intense productivity events are not observed during the SW intermonsoon, but rather during the NE intermonsoon. During the intermonsoon redox elements in the Western portion of the basin appear to be more oxidized relative to the in situ production.

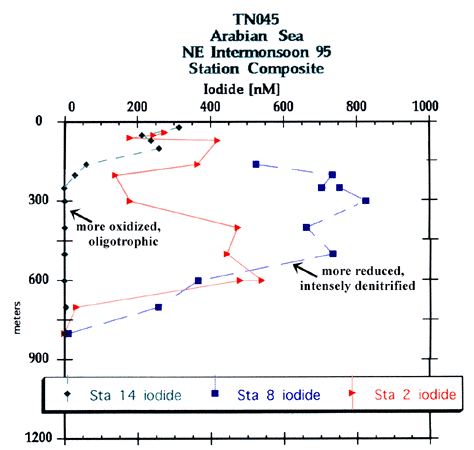
Above, in the 1992/1995 Composite figure shown here. Iodine in the upper 400 m is found to be in a more reduced state during the NE intermonsoon and in a more oxidized state during the SW intermonsoon. During the Southwest monsoon the iodide in the top 100 m is more reduced than the SW intermonsoon while more oxidized than the NE intermonsoon and more reduced than both, between 250-400 m. All three stations were sampled in 3200 m of water off the coast of Oman.
These two stations were sampled at high resolution (5 m-10 m) through the OMZ. They were sampled with multiple casts (24 depths) per cast given the resolution of the NO2-, I have plotted each cast with a different symbol and color. More samples await analysis and will increase the resolution with some depths being duplicated from cast to cast.
Discussion of the Arabian Sea data
Iodide concentration proportional to productivity
In collaboration with Tim Jickells and Lucia Campos at the University of East Anglia, we have pooled our time-series data sets from ALOHA and BATS and discovered that the integrated iodide concentrations at the two time-series stations are proportional to a yearly averaged production value unique to each station (Campos et al., 1996).
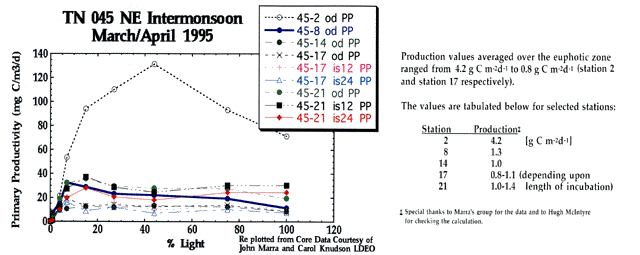
Productivity values from Process #2
A wide range of productivity values were obtained during the Process #2 survey. The corresponding iodide profiles are shown below along side productivity data provided by Carol Knudson and John Marra of Lamont Doherty Earth Observatory. Iodide speciation reflects both productivity differences and water column denitrification which contributes to the reduction of iodate.
Iodide oxidation, kinetically swift
As a consequence, we consider the OMZ (water column) of the Arabian Sea as comparable to a recent sedimentary environment with redox fronts.
Accumulation of I- as seen in parts of the Arabian Basin, suggests a diagenetic cycling mechanism. Kennedy and Elderfield (1987) reported I- released in (pelagic) porewaters followed by rapid removal of I-onto organic matter and I/C values up to 200 X 10 -4. A sedimentary framework allows for the accumulation of I- in the eastern portion of the basin. The search for viable oxidants in the OMZ of the Arabian Sea continues.
Significant Questions to be Addressed:
- What are the most abundant oxidants available in this productive system?
- Can we determine (rather than infer) the mechanisms of the redox couple I-/IO3- ?
- Will we be able to apply similar enzyme systems to nitrogen species as well?
- Work in progress includes the study of the reduction of IO3- in culture and oxidation of I- in the presence of peroxidase, H2O2 and/or OCl-.
Preliminary experimental data: Biotic and Abiotic mechanisms of iodate reduction
- Experimental data show that Shewanella Putrefaciens (MR-4), a species closely related to Pseudomonas/Alteromonas identified in the Arabian Sea by Nair et al. (1994) is capable of reducing iodate in culture. Moreover, the carbon substrate alone, (in this case: lactate) is capable of reducing (~ 10%) of the iodate added to the artificial seawater (Farrenkopf et al, in prep).
- Experimental data also show that ammonia (in the form NH4Cl) is capable of reducing iodate in artificial seawater (Farrenkopf, unpubl.).
Concluding Remarks
The iodide profiles shown here from the 1995 NE intermonsoon transect are significantly different from the sub-surface iodide maxima shown from the 1992 SW intermonsoon transect data. The most recent data set shows much more reduction taking place in the water column; whether this is due to inter-annual or to seasonal differences cannot be ascertained with the data in hand. However, based upon time-series data we suspect that these features reflect seasonal differences which include fast re oxidation kinetics (orders of weeks to months). The quest continues for strong (or efficient) oxidants in low oxygen/non-sulfidic water column environments.
References
Brandão, Ana Claudia M.; Angela de Luca Rebello Wagener; and Klaus Wagener (1994) Model experiments on the diurnal cycling of iodine in seawater. Marine Chemistry 46: 25-31.
Campos, M.L.A.; A.M. Farrenkopf; T. D. Jickells and G.W. Luther, III (1996) A comparison of dissolved iodine cycling at the Bermuda Atlantic Time Series Station and the Hawaii Ocean Time-series. Deep Sea Research II 43; 455-63.
Farrenkopf, Anna M. (1993) "Vertical Distribution of Dissolved Iodine Species in the Northwest Indian Ocean." M.S., University of Delaware.
Farrenkopf, A.M.; G.W. Luther, III; V.W. Truesdale; and C.H. van der Weijden (in press DSR Special Volume) Sub-surface iodide maxima: Evidence for biologically catalyzed redox cycling in Arabian Sea OMZ during the SW intermonsoon.
Luther, George W., III (1991) Sulfur and iodine speciation in the water column of the Black Sea, in Black Sea Oceanography, E. Izdar and J. W. Murray, Editors. Kluwer Publishers: Netherlands. p. 187-204.
Nair, Shanta; Ponnampakam A. Loka Bharathi; and Dorairajsingham Chandramohan (1994) Cultureable bacteria from the euphotic zone of the Indian Ocean during the summer monsoon. Oceanologica Acta, 17(1): 63-68.
Tsunogai, S. and T. Sase (1969) Formation of iodide-iodine in the ocean. Deep-Sea Research, 16: 489-496.
Wong, George T.F.; Kazufumi Takayanagi; and James F. Todd, (1985) Dissolved iodine in waters overlying and in the Orca Basin, Gulf of Mexico. Marine chemistry, 17: 177-183.
Wong, George T.F. and Peter G. Brewer (1977) The marine chemistry of iodine in anoxic basins. Geochimica et Cosmochimica Acta, 41: 151-159.
Acknowledgments:
We thank Captain Gomes and the crew of the R/V Thompson; the technicians from University of Washington; and all the techs and PI's that consulted and provided data presented here today. This work was made possible by two NSF grants provided to GWL3 (OCE-9217245 and OCE-9310792). Revisiting the work of Hilary Kennedy, and invaluable discussions with Tim Jickells, Allison Butler, and Hugh MacIntyre, are helping with the interpretation of this new data. Thanks are due to Kelley Warren, Steve Theberge, Katherine A. Bouton and Mary Zawoysky for aiding with the preparation of this web formatted poster.
Availability of data was definitely an asset and a credit to the U.S. JGOFS PI's that have allowed access to data generated during this Arabian Sea Process cruise. Special thanks to John Marra, Chief Scientist and Lou Codispoti for continuing discussions.